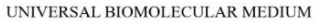
| Serial Number | 79436392 |
| Word Mark | UNIVERSAL BIOMOLECULAR MEDIUM |
| Filing Date | Monday, May 12, 2025 |
| Status | 645 - FINAL REFUSAL - MAILED |
| Status Date | Tuesday, May 12, 2026 |
| Registration Number | 0000000 |
| Registration Date | NOT AVAILABLE |
| Mark Drawing | 4 - Illustration: Drawing with word(s) / letter(s) / number(s) in Block form |
| Published for Opposition Date | NOT AVAILABLE |
| Goods and Services | Chemical preparations and chemical substances other than for medical or veterinary purposes, namely, for use in laboratory, analytical, diagnostic, clinical or medical applications, in particular for the preparation of media for the preservation and transport of DNA, RNA and proteins for the analysis of viruses, bacteria and other microorganisms; chemicals, chemical substances and molecules, and combinations thereof, other than for medical or veterinary purposes, namely, intended to facilitate chemical, molecular and biochemical analysis of biological and chemical samples and substances collected in a preservation and transport medium, all the aforesaid goods only for use with nasopharyngeal samples and urine and only for the analysis of respiratory viruses and Gram-positive bacteria, Gram-negative bacteria and fungi, to the exclusion of all other samples, microorganisms and uses |
| Goods and Services | Disposable devices, namely storage tubes and swabs for the collection, preservation and transport of biological and chemical samples and substances, not for medical use; scientific research specimen tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, not for medical use; scientific research specimen tubes containing lysing and inactivating media for viruses, bacteria and other microorganisms, intended to preserve their components and macromolecules, not for medical use; scientific research specimen tubes containing media intended to preserve components and macromolecules of viruses, bacteria and other microorganisms, not for medical use; flocked swabs for the collection, preservation and transport of viruses, bacteria and other microorganisms and their components, not for medical use; swabs for the collection, preservation and transport of viruses, bacteria and other microorganisms and their components, not for medical use; containers and kits comprised primarily of sample vials and tubes for the preservation and transport of biological, chemical and diagnostic samples, not for medical use; containers and kits comprised primarily of sample vials and tubes for the lysis and inactivation of viruses, bacteria, other microorganisms and cells, not for medical use; containers and kits comprised primarily of sample vials and tubes for preserving components and macromolecules of viruses, bacteria and other microorganisms, not for medical use; kits comprising flocked swabs and scientific research specimen tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, not for medical use; kits comprising swabs and scientific research specimen tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, not for medical use; kits comprising flocked swabs and scientific research specimen tubes containing lysing and inactivating media for viruses, bacteria and other microorganisms, intended to preserve their components and macromolecules, not for medical use; kits comprising swabs and scientific research specimen tubes containing lysing and inactivating media for viruses, bacteria and other microorganisms, intended to preserve their components and macromolecules, not for medical use; kits comprising flocked swabs and scientific research specimen tubes containing media for preserving components and macromolecules of viruses, bacteria and other microorganisms, not for medical use; kits comprising swabs and scientific research specimen tubes containing media for preserving components and macromolecules of viruses, bacteria and other microorganisms, not for medical use, all the aforesaid goods only for use with nasopharyngeal samples and urine and only for the analysis of respiratory viruses and Gram-positive bacteria, Gram-negative bacteria and fungi, to the exclusion of all other samples, microorganisms and uses |
| Goods and Services | Disposable devices, namely, medical specimen tubes and swabs for the collection, preservation and transport of biological and chemical samples and substances, for medical use; medical specimen collection tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, for medical use; medical specimen collection tubes containing lysing and inactivating media for viruses, bacteria and other microorganisms, intended to preserve their components and macromolecules, for medical use; medical specimen collection tubes containing media for preserving components and macromolecules of viruses, bacteria and other microorganisms, for medical use; containers and kits comprised primarily of medical sample vials and tubes for the transport and preservation of biological, chemical and diagnostic samples, for medical use; containers and kits comprised primarily of medical sample vials and tubes for the lysis and inactivation of viruses, bacteria, other microorganisms and cells, for medical use; containers and kits comprised primarily of medical sample vials and tubes for preserving components and macromolecules of viruses, bacteria and other microorganisms, for medical use; kits comprising medical sample vials and tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, for medical use; kits comprising medical sample vials and tubes containing preservation and transport media for viruses, bacteria and other microorganisms and their components, for medical use; kits comprising medical sample vials and tubes containing lytic and inactivating media for viruses, bacteria and other microorganisms intended to preserve their components and macromolecules, for medical use; kits comprising medical sample vials and tubes containing media intended to preserve components and macromolecules of viruses, bacteria and other microorganisms, for medical use, all the aforesaid goods only for use with nasopharyngeal samples and urine and only for the analysis of respiratory viruses and Gram-positive bacteria, Gram-negative bacteria and fungi, to the exclusion of all other samples, microorganisms and uses |
| International Class | 001 - Chemicals used in industry, science and photography, as well as in agriculture, horticulture and forestry; unprocessed artificial resins; unprocessed plastics; manures; fire extinguishing compositions; tempering and soldering preparations; chemical substances for preserving foodstuffs; tanning substances; adhesives used in industry. |
| US Class Codes | 001, 005, 006, 010, 026, 046 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 001 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 009 - Scientific, nautical, surveying, photographic, cinematographic, optical, weighing, measuring, signalling, checking (supervision), life-saving and teaching apparatus and instruments; apparatus and instruments for conducting, switching, transforming, accumulating, regulating or controlling electricity; apparatus for recording, transmission or reproduction of sound or images; magnetic data carriers, recording discs; automatic vending machines and mechanisms for coin operated apparatus; cash registers, calculating machines, data processing equipment and computers; fire extinguishing apparatus. |
| US Class Codes | 021, 023, 026, 036, 038 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 009 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 010 - Surgical, medical, dental, and veterinary apparatus and instruments, artificial limbs, eyes, and teeth; orthopedic articles; suture materials. |
| US Class Codes | 026, 039, 044 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 010 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| Party Name | COPAN ITALIA S.p.A. |
| Party Type | 10 - Original Applicant |
| Legal Entity Type | 99 - Other |
| Address | IT |
| Event Date | Event Description |
| Thursday, November 6, 2025 | SN ASSIGNED FOR SECT 66A APPL FROM IB |
| Thursday, November 6, 2025 | NEW APPLICATION OFFICE SUPPLIED DATA ENTERED |
| Thursday, November 6, 2025 | APPLICATION FILING RECEIPT MAILED |
| Friday, November 7, 2025 | ASSIGNED TO EXAMINER |
| Wednesday, November 12, 2025 | NON-FINAL ACTION WRITTEN |
| Thursday, November 13, 2025 | NON-FINAL ACTION (IB REFUSAL) PREPARED FOR REVIEW |
| Friday, December 19, 2025 | REFUSAL PROCESSED BY MPU |
| Friday, December 19, 2025 | NON-FINAL ACTION MAILED - REFUSAL SENT TO IB |
| Thursday, January 8, 2026 | REFUSAL PROCESSED BY IB |
| Tuesday, April 7, 2026 | TEAS RESPONSE TO OFFICE ACTION RECEIVED |
| Tuesday, April 7, 2026 | CORRESPONDENCE RECEIVED IN LAW OFFICE |
| Tuesday, April 7, 2026 | TEAS/EMAIL CORRESPONDENCE ENTERED |
| Tuesday, May 12, 2026 | FINAL REFUSAL WRITTEN |
| Tuesday, May 12, 2026 | FINAL REFUSAL E-MAILED |
| Tuesday, May 12, 2026 | NOTIFICATION OF FINAL REFUSAL EMAILED |