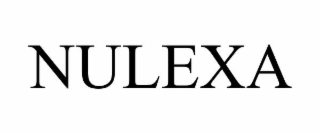
| Serial Number | 99483119 |
| Word Mark | NULEXA |
| Filing Date | Thursday, November 6, 2025 |
| Status | 686 - PUBLISHED FOR OPPOSITION |
| Status Date | Tuesday, May 12, 2026 |
| Registration Number | 0000000 |
| Registration Date | NOT AVAILABLE |
| Mark Drawing | 4 - Illustration: Drawing with word(s) / letter(s) / number(s) in Block form |
| Published for Opposition Date | Tuesday, May 12, 2026 |
| Goods and Services | Medical diagnostic apparatus for testing and analyzing blood and other bodily fluids; Medical diagnostic apparatus for testing for viruses; Diagnostic kits consisting primarily of probes, buffers and reagents for use in microbial testing; Sample preparation device for medical diagnostic uses; Medical diagnostic apparatus for testing blood and other bodily fluids incorporating recorded operating system software; Medical diagnostic apparatus for detecting viruses and bacteria with embedded operating Software in a Medical Device (SiMD); Medical diagnostic apparatus for detecting viruses and bacteria |
| Goods and Services | Medical services, namely, medical molecular diagnostic, testing, monitoring, and reporting services |
| Goods and Services | Medical diagnostic test media in the form of a cartridge for measuring, testing and analyzing blood and other bodily fluids for use in the detection of infectious diseases, including viral and bacterial pathogens; Diagnostic reagents for medical use in in vitro diagnosis; In vitro diagnostic preparations for medical use; In vitro diagnostic preparations for medical purposes; Medical diagnostic reagents and assays for testing of body fluids; Diagnostic kits comprised of medical diagnostic reagents and assays for testing of bodily fluids for use in disease detection, namely, viral and bacterial pathogens; Diagnostic kits comprised of medical diagnostic reagents and assays for testing of bodily fluids for use in disease detection, namely, Influenza A, Influenza B, SARS-CoV-2, Respiratory Syncytial Virus (RSV), and Group A Streptococcus (Strep A); Diagnostic kits comprising diagnostic agents, preparations and substances for medical purposes; Diagnostic kits comprising diagnostic agents, preparations and substances for medical use; Diagnostic test reagents for medical use; Diagnostic test reagents for medical purposes; Medical diagnostic reagents; Medical diagnostic reagents for the analysis of body fluids; Diagnostic chemical reagents for medical use; Diagnostic chemical reagents for medical purposes |
| International Class | 005 - Pharmaceutical and veterinary preparations; sanitary preparations for medical purposes; dietetic substances adapted for medical use, food for babies; plasters, materials for dressings; material for stopping teeth, dental wax; disinfectants; preparations for destroying vermin; fungicides, herbicides. |
| US Class Codes | 005, 006, 018, 044, 046, 051, 052 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 005 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 010 - Surgical, medical, dental, and veterinary apparatus and instruments, artificial limbs, eyes, and teeth; orthopedic articles; suture materials. |
| US Class Codes | 026, 039, 044 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 010 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 044 - Medical services; veterinary services; hygienic and beauty care for human beings or animals; agriculture, horticulture and forestry services. |
| US Class Codes | 100, 101 |
| Class Status Code | 6 - Active |
| Class Status Date | Thursday, November 6, 2025 |
| Primary Code | 044 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| Party Name | Ortho-Clinical Diagnostics, Inc. |
| Party Type | 20 - Owner at Publication |
| Legal Entity Type | 03 - Corporation |
| Address | Raritan, NJ 08869 US |
| Party Name | Ortho-Clinical Diagnostics, Inc. |
| Party Type | 10 - Original Applicant |
| Legal Entity Type | 03 - Corporation |
| Address | Raritan, NJ 08869 US |
| Event Date | Event Description |
| Thursday, November 6, 2025 | NEW APPLICATION ENTERED |
| Thursday, November 6, 2025 | APPLICATION FILING RECEIPT MAILED |
| Wednesday, February 11, 2026 | NEW APPLICATION OFFICE SUPPLIED DATA ENTERED |
| Wednesday, March 18, 2026 | ASSIGNED TO EXAMINER |
| Thursday, April 2, 2026 | NON-FINAL ACTION WRITTEN |
| Thursday, April 2, 2026 | NON-FINAL ACTION E-MAILED |
| Thursday, April 2, 2026 | NOTIFICATION OF NON-FINAL ACTION E-MAILED |
| Tuesday, April 7, 2026 | TEAS RESPONSE TO OFFICE ACTION RECEIVED |
| Tuesday, April 7, 2026 | CORRESPONDENCE RECEIVED IN LAW OFFICE |
| Tuesday, April 7, 2026 | TEAS/EMAIL CORRESPONDENCE ENTERED |
| Wednesday, April 15, 2026 | APPROVED FOR PUB - PRINCIPAL REGISTER |
| Wednesday, May 6, 2026 | NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED |
| Tuesday, May 12, 2026 | PUBLISHED FOR OPPOSITION |
| Tuesday, May 12, 2026 | OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED |