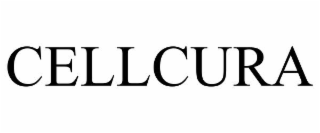
| Serial Number | 99302295 |
| Word Mark | CELLCURA |
| Filing Date | Friday, July 25, 2025 |
| Status | 688 - NOTICE OF ALLOWANCE - ISSUED |
| Status Date | Tuesday, April 21, 2026 |
| Registration Number | 0000000 |
| Registration Date | NOT AVAILABLE |
| Mark Drawing | 4 - Illustration: Drawing with word(s) / letter(s) / number(s) in Block form |
| Published for Opposition Date | Tuesday, February 24, 2026 |
| Goods and Services | Medical cleansers for skin and wounds; Liquid bandages for skin wounds; Pharmaceutical preparations for skin wounds; Bandages for skin wounds; Pharmaceutical preparations for wounds; Biological preparations for the treatment of musculoskeletal and orthopedic conditions including osteoarthritis, tendon and ligament injuries, fractures and non-unions, bulging or herniated discs, degenerative disc disease, carpal tunnel syndrome, cartilage injuries, muscle injuries, temporomandibular joint (TMJ) disorder, piriformis syndrome, sciatica, sacroiliac (SI) joint dysfunction, and osteonecrosis; Biological preparations for the treatment of autoimmune and inflammatory conditions including multiple sclerosis (MS), Crohn’s disease, and rheumatoid arthritis; Biological preparations for the treatment of neurological conditions including stroke, dementia, Parkinson’s disease, Huntington’s disease, spinal cord injury, post-concussion syndrome, and autism spectrum disorder; Biological preparations for the treatment of cardiovascular diseases including heart failure, peripheral artery disease, ischemic heart disease, and general cardiovascular disease; Biological preparations for the treatment of metabolic diseases including Type 1 and Type 2 diabetes, and obesity; Biological preparations for the treatment of hematological and immune diseases including, leukemia, lymphoma, myeloma, myelodysplastic syndrome, and myeloproliferative disorders; Biological preparations for the treatment of ophthalmologic conditions including macular degeneration, retinal detachment, and macular hole; Biological preparations for the treatment of liver and gastrointestinal diseases including chronic liver diseases, cirrhosis, liver failure, and Inflammatory bowel disease; Biological preparations for the treatment of chronic pain and degenerative conditions including fibromyalgia, chronic joint pain, and chronic wounds or non-healing ulcers |
| International Class | 005 - Pharmaceutical and veterinary preparations; sanitary preparations for medical purposes; dietetic substances adapted for medical use, food for babies; plasters, materials for dressings; material for stopping teeth, dental wax; disinfectants; preparations for destroying vermin; fungicides, herbicides. |
| US Class Codes | 005, 006, 018, 044, 046, 051, 052 |
| Class Status Code | 6 - Active |
| Class Status Date | Friday, July 25, 2025 |
| Primary Code | 005 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| Party Name | Cellcura Sciences, Inc. |
| Party Type | 20 - Owner at Publication |
| Legal Entity Type | 03 - Corporation |
| Address | Lehi, UT 84043 US |
| Party Name | Cellcura Sciences, Inc. |
| Party Type | 10 - Original Applicant |
| Legal Entity Type | 03 - Corporation |
| Address | Lehi, UT 84043 US |
| Event Date | Event Description |
| Friday, July 25, 2025 | NEW APPLICATION ENTERED |
| Friday, July 25, 2025 | APPLICATION FILING RECEIPT MAILED |
| Friday, July 25, 2025 | NEW APPLICATION OFFICE SUPPLIED DATA ENTERED |
| Thursday, December 11, 2025 | ASSIGNED TO EXAMINER |
| Friday, December 12, 2025 | NON-FINAL ACTION WRITTEN |
| Friday, December 12, 2025 | NON-FINAL ACTION E-MAILED |
| Friday, December 12, 2025 | NOTIFICATION OF NON-FINAL ACTION E-MAILED |
| Tuesday, January 20, 2026 | TEAS RESPONSE TO OFFICE ACTION RECEIVED |
| Tuesday, January 20, 2026 | CORRESPONDENCE RECEIVED IN LAW OFFICE |
| Tuesday, January 20, 2026 | TEAS/EMAIL CORRESPONDENCE ENTERED |
| Monday, February 2, 2026 | APPROVED FOR PUB - PRINCIPAL REGISTER |
| Wednesday, February 18, 2026 | NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED |
| Tuesday, February 24, 2026 | PUBLISHED FOR OPPOSITION |
| Tuesday, February 24, 2026 | OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED |
| Tuesday, April 21, 2026 | NOA E-MAILED - SOU REQUIRED FROM APPLICANT |