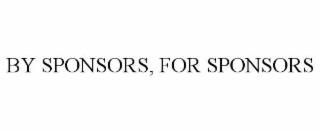
| Serial Number | 99282560 |
| Word Mark | BY SPONSORS, FOR SPONSORS |
| Filing Date | Monday, July 14, 2025 |
| Status | 688 - NOTICE OF ALLOWANCE - ISSUED |
| Status Date | Tuesday, February 24, 2026 |
| Registration Number | 0000000 |
| Registration Date | NOT AVAILABLE |
| Mark Drawing | 4 - Illustration: Drawing with word(s) / letter(s) / number(s) in Block form |
| Published for Opposition Date | Tuesday, December 30, 2025 |
| Goods and Services | Business management and consultation in the field of pharmaceutical, biotechnology, medical devices, and life sciences on behalf of pharmaceutical, biotechnology, medical devices and life sciences companies to assist them with clinical research, clinical trials and applications for regulatory approval; Business management and consultation in the field of drugs, biologics, and medical devices research and development and manufacturing on behalf of pharmaceutical, biotechnology, medical devices, and life sciences companies to assist them with clinical research, clinical trials, and applications for regulatory approval; Business management and consultation in the field of information technology regulatory compliance, namely, providing information management services; Outsourcing services in the nature of arranging service contracts for others in the field of pharmaceutical, biotechnology, medical devices, and life sciences; Outsourcing services in the nature of arranging service contracts for others in the field of drugs, biologics, and medical devices research and development and manufacturing; Recruitment and placement of personnel in the field of pharmaceutical, biotechnology, medical devices, and life sciences; Business assistance, advisory and consulting services in the field of pharmaceutical, biotechnology, medical devices, and life sciences; Business assistance, advisory and consulting services in the field of drugs, biologics, medical devices, and life sciences research and development and manufacturing; Business data analysis services in the field of pharmaceutical, biotechnology, medical devices, and life sciences; Business data analysis services in the field of drugs, biologics, medical devices, and life sciences research and development and manufacturing; Professional staffing services in the field of pharmaceutical, biotechnology, medical devices, and life sciences |
| Goods and Services | Technological planning and consulting services in the field of life sciences; Providing information in the field of clinical research via a website; Providing a website featuring educational information in the field of clinical research; Medical and scientific research, namely, conducting clinical trials for others; Consulting services for others in the field of design, planning, and implementation project management of medical and scientific research and clinical trials; Consulting services for others in the field of design, planning, and implementation project management of clinical trials in the fields of pharmaceutical, biotechnology, medical devices, and life sciences; Quality management services, namely, quality evaluation and analysis, quality assurance, and quality control, in the field of biotechnology; Quality management services, namely, quality evaluation and analysis, quality assurance, and quality control, in the field of pharmaceutical; Quality management services, namely, quality evaluation and analysis, quality assurance, and quality control, in the field of medical devices; Quality management services, namely, quality evaluation and analysis, quality assurance, and quality control, in the field of life sciences; Consulting services for others in the field of design, planning, and implementation project management of information technology and information management services in the nature of life sciences; Consulting services in the field of pharmaceutical research and development; Consulting services in the field of biotechnology; Scientific research in the nature of conducting clinical trials for others in the field of pharmaceutical, biotechnology, medical devices, and life sciences; Providing medical and scientific research information in the field of pharmaceuticals and clinical trials; Scientific and technological services, namely, research and design in the field of pharmaceutical , biotechnology, medical devices, and life sciences, namely, conducting and analyzing clinical trials |
| Goods and Services | Regulatory compliance consultancy in the field of biotechnology; Regulatory compliance consulting in the field of medical devices; Regulatory compliance consulting in the field of life sciences; Regulatory compliance consulting in the field of information technology and information management services; Regulatory compliance consulting in the field of pharmaceutical; Regulatory compliance consulting in the field of pharmaceutical, biotechnology, medical devices, and life sciences, in the nature of health authority inspection readiness and remediation |
| Goods and Services | Educational services, namely, conducting webinars and live workshops, providing classes and training programs, conducting seminars and conferences, providing online non-downloadable videos, and providing webcasts and podcasts in the field of pharmaceutical, biotechnology, medical devices, and life sciences and distribution of training material in connection therewith; Providing on-line newsletters in the field of pharmaceutical, biotechnology, medical devices and life sciences; Educational services, namely, conducting webinars and live workshops, providing classes and training programs, conducting seminars and conferences, providing online non-downloadable videos, and providing webcasts and podcasts in the field of drugs, biologics, medical devices, life sciences regulatory compliance, research and development, and manufacturing and distribution of training material in connection therewith; On-line journals, namely, blogs featuring information in the field of pharmaceutical, biotechnology, medical devices and life sciences; Providing on-line newsletters in the field of drugs, biologics, medical devices, life sciences regulatory compliance, research and development, and manufacturing |
| International Class | 035 - Advertising; business management; business administration; office functions. |
| US Class Codes | 100, 101, 102 |
| Class Status Code | 6 - Active |
| Class Status Date | Monday, July 14, 2025 |
| Primary Code | 035 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 041 - Education; providing of training; entertainment; sporting and cultural activities. |
| US Class Codes | 100, 101, 107 |
| Class Status Code | 6 - Active |
| Class Status Date | Monday, July 14, 2025 |
| Primary Code | 041 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 042 - Scientific and technological services and research and design relating thereto; industrial analysis and research services; design and development of computer hardware and software. |
| US Class Codes | 100, 101 |
| Class Status Code | 6 - Active |
| Class Status Date | Monday, July 14, 2025 |
| Primary Code | 042 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| International Class | 045 - Legal services; security services for the protection of property and individuals; personal and social services rendered by others to meet the needs of individuals. |
| US Class Codes | 100, 101 |
| Class Status Code | 6 - Active |
| Class Status Date | Monday, July 14, 2025 |
| Primary Code | 045 |
| First Use Anywhere Date | NOT AVAILABLE |
| First Use In Commerce Date | NOT AVAILABLE |
| Party Name | GALAXIGEN, LLC |
| Party Type | 20 - Owner at Publication |
| Legal Entity Type | 16 - Limited Liability Company |
| Address | Palm Harbor, FL 34684 US |
| Party Name | GALAXIGEN, LLC |
| Party Type | 10 - Original Applicant |
| Legal Entity Type | 16 - Limited Liability Company |
| Address | Palm Harbor, FL 34684 US |
| Event Date | Event Description |
| Monday, July 14, 2025 | NEW APPLICATION ENTERED |
| Monday, July 14, 2025 | APPLICATION FILING RECEIPT MAILED |
| Tuesday, November 18, 2025 | NEW APPLICATION OFFICE SUPPLIED DATA ENTERED |
| Tuesday, December 2, 2025 | ASSIGNED TO EXAMINER |
| Tuesday, December 2, 2025 | NON-FINAL ACTION WRITTEN |
| Tuesday, December 2, 2025 | NON-FINAL ACTION E-MAILED |
| Tuesday, December 2, 2025 | NOTIFICATION OF NON-FINAL ACTION E-MAILED |
| Wednesday, December 3, 2025 | TEAS RESPONSE TO OFFICE ACTION RECEIVED |
| Wednesday, December 3, 2025 | CORRESPONDENCE RECEIVED IN LAW OFFICE |
| Wednesday, December 3, 2025 | TEAS/EMAIL CORRESPONDENCE ENTERED |
| Thursday, December 4, 2025 | APPROVED FOR PUB - PRINCIPAL REGISTER |
| Wednesday, December 24, 2025 | NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED |
| Tuesday, December 30, 2025 | PUBLISHED FOR OPPOSITION |
| Tuesday, December 30, 2025 | OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED |
| Tuesday, February 24, 2026 | NOA E-MAILED - SOU REQUIRED FROM APPLICANT |